
The A.i.r. Platform™ from MGS represents a new era in customizable auto-injector platforms. Built around a flexible core technology, it enables Pharmaceutical partners to develop bespoke, patient-centered devices faster and more cost effectively. Below, we answer the most common questions about how the A.i.r. Platform™ works and why it’s redefining what’s possible in combination product development and manufacturing.
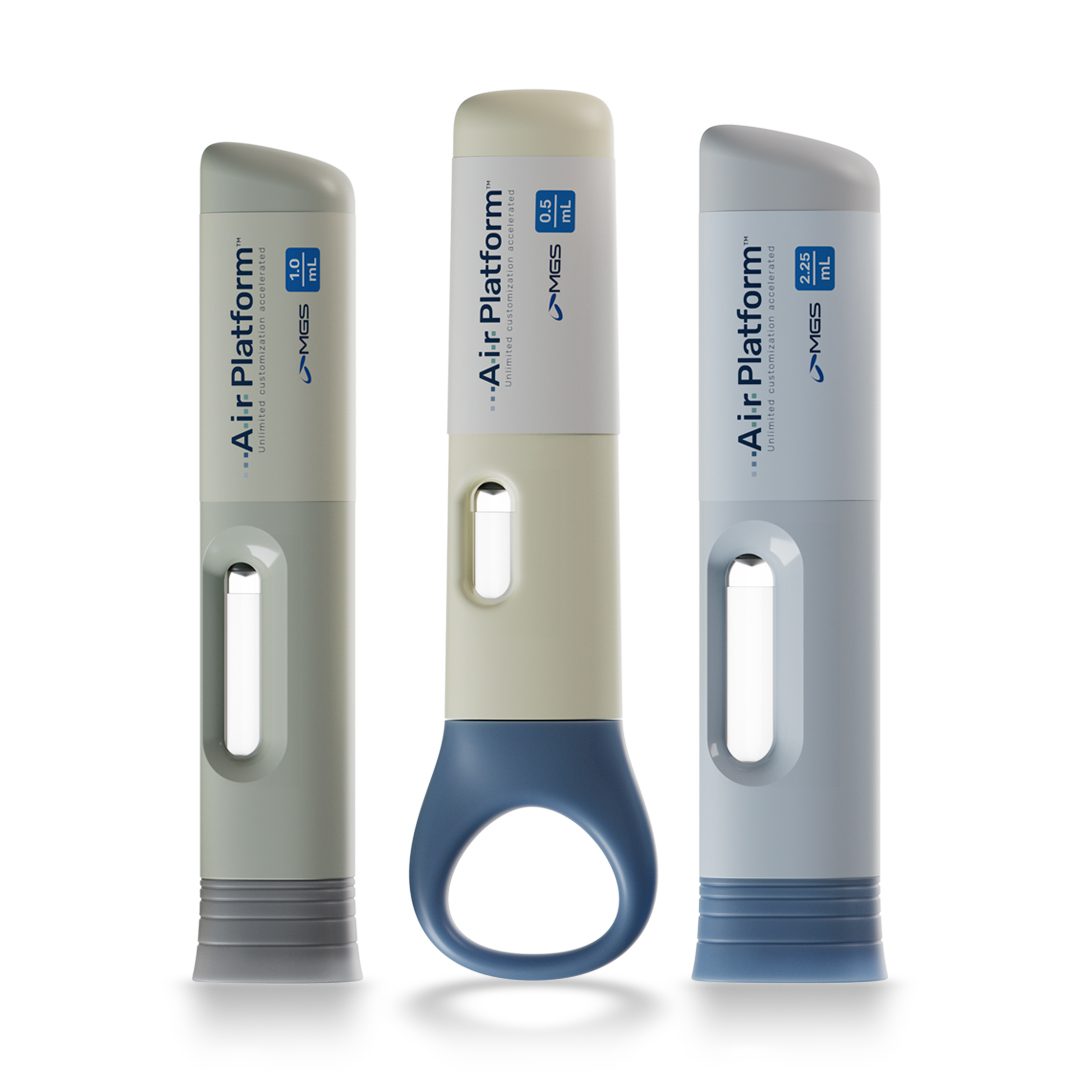
Can auto-injectors like the A.i.r. Platform™ be customized for specific patient populations?
Yes, the A.i.r. Platform™ enables you to customize the device to fit your patient needs. In a structured process, MGS Design & Development works with you to design the physical appearance of the device to meet unique patient needs. Our focus is on ensuring patients in your focus area can use the device as intended, even if they are challenged with lower dexterity or low strength. Our team looks at outer shape, surfaces, cap design and adjustments in force – both for activation and hold force – to meet specific needs.
The A.i.r. Platform™ also offers continuous audible feedback as an option. By implementing this feature, you are able to improve feedback to the user during injection, reducing risk from early lifts that cause underdosing.
The usability documentation and risk assessment was developed by the MGS team, built on an understanding of 14 typical diseases within subcutaneous drug delivery. Documentation from this platform can easily be adapted to your disease area, saving additional time and cost in finalizing instructions and documentation.
Can the A.i.r. Platform™ be customized to accommodate different drug profiles, including high viscosity and varying fill volumes?
The A.i.r. Platform™ can be customized to accommodate different drug profiles, with key characteristics including viscosity, dose volume and associated injection time requirements. The A.i.r. Platform is designed to accommodate these variables, supporting a wide range of viscosities and fill volumes from 0.3 mL to 2.25 mL. The A.i.r. Platform™ is also customizable to fit any desired Pre-Filled Syringe (PFS).
The following outlines how these key characteristics can be accommodated within the A.i.r. Platform™.
| Drug Profile Requirement | How the A.i.r. Platform™ Addresses It | Supported Range |
|---|---|---|
| Drug viscosity | Power spring and device parts can be customized to support any viscosity within our range. | Suitable for low- to high-viscosity formulations (0-200 cP). |
| Fill volumes | Configurable platform supports multiple dose volumes for any PFS within our fill volume range. | 0.3 mL to 2.25 mL fill volumes. |
| Injection time | Customizable power spring and device parts meet any injection time requirements. | Adjustable to align with drug and patient needs. Supported range: 1-20 seconds. |
| Pre-filled syringe (PFS) use | Designed to work with any PFS, including cartridges. | Supports any PFS formats and brand within 0.3mL-2.25 mL. |
Is the A.i.r. Platform™ compatible with different prefilled syringe brands and formats?
The A.i.r. Platform™ can be customized to any Pre-Filled Syringe (PFS) within the platform’s fill volume of 0.3 to 2.25 mL. If desired, the platform can also be customized to accommodate a cartridge in place of a PFS.
The A.i.r. Platform™ follows this approach by allowing alignment with any PFS brand and formats without requiring a complete device redesign.
Can the A.i.r. Platform™ be customized to reflect brand identity while meeting usability and regulatory requirements?
The A.i.r Platform™ is fully customizable to reflect your unique brand identity. Our designers work with your team to evaluate shape, color(s), embossments and more, helping you build your own device brand to support product marketing efforts.
The look and feel of your auto-injector should reflect both your brand and the patients it serves. Thoughtful design elements like shape, color and finish can help create a more recognizable and cohesive product experience, while reinforcing your understanding of patient needs. Customization also creates opportunities to align with broader brand priorities, such as sustainability or therapy-specific positioning.
In practice, brand expression is typically achieved through changes to visible and tactile elements of the device, while preserving core functionality and validated use conditions. This approach allows companies to differentiate their device appearance without compromising usability or regulatory compliance.
The A.i.r. Platform™ supports this approach by allowing brand-specific design elements to be adapted within an established platform framework.
Brand-Related Elements That Can Be Customized
| Design Element | Customization Scope |
|---|---|
| Device color | The device color can be customized to fit company and/or drug brand as desired. |
| Surface finish | Surfaces can be customized to reflect design DNA and/or to support user interface with device. |
| Body shape | The external form factor (shape and size) of the device can be fully designed to represent company design DNA and be designed around specific user needs. |
| Embossments | Logos, symbols and orientation cues can be implemented as desired. |
| Injection feedback | The device can be designed to give needed feedback to the specific user population including both audible and visual cues – i.e., continuous feedback. |
How can MGS’ A.i.r. Platform™ reduce product development timelines?
With MGS’ A.i.r. Platform™, you can reduce development timelines by allowing product design and development teams to build on an established foundation rather than designing a device from scratch.
This approach shortens development time by:
- Building on a platform, reducing early design and feasibility work.
- Customizing your device on already filed patents for the platform.
- Focusing customization on defined areas related to the drug, patient population or brand.
- Reducing time to first devices in-hand as you utilize existing core technology.
- Accelerate timelines by leveraging existing usability documentation.
As a result, development efforts can move more efficiently from concept to clinical trial and commercial readiness.
How can Pharmaceutical companies reduce development and regulatory risk when developing an auto-injector?
Pharmaceutical companies can reduce development and regulatory risk when developing an auto-injector by relying on established device approaches and experienced partners, rather than creating entirely new systems.
Through the A.i.r. Platform™, MGS helps you reduce risk with a proven development framework, including usability documentation and completed risk assessments developed from an understanding of 14 disease areas. Leveraging the expertise of MGS’ experienced Design & Development teams, Pharma innovators are able to focus on the drug itself while reducing uncertainty and rework associated with drug delivery device design, testing and manufacturing.
Together, these practices help streamline regulatory interactions and improve the likelihood of successful approval.
What are the key steps to developing our own auto-injector platform?
Developing and manufacturing an in-house auto-injector platform typically follows a staged process that builds capacity over time. At MGS, we often begin with a two-week feasibility sprint to evaluate concepts and reduce early risk. From there, development progresses toward initial prototype devices, with timelines tailored to your program and needs.
Key steps include:
- Define the platform scope, including target drug profiles, patient populations and anticipated future programs.
- Select or define a baseline device concept that can be adapted across multiple applications.
- Incorporate usability and human-factors requirements early, informed by patient and clinician needs.
- Deliver functional prototypes for early testing, followed by finalized designs and complete functional devices.
- Execute verification and validation activities in alignment with regulatory expectations.
- Prepare for manufacturing scale-up and lifecycle management, including Tooling, Automation and Manufacturing considerations.
Taking a structured, stepped approach helps teams manage complexity while supporting future expansion of the platform.
How do you help Pharmaceutical companies reduce auto-injector development costs?
Pharmaceutical companies can reduce auto-injector development costs by working with MGS to customize the A.i.r. Platform™ to their unique drug, brand and patient needs. Because MGS has already invested years in developing this platform, that time and cost are removed from your auto-injector development timeline, helping you reach the market faster and more efficiently.
This approach can also support lower complaint rates once your combination product is on the market. When complaints occur, they often trigger investigations, product replacements and additional follow-up activities such as issue documentation and customer communications. Reducing these events helps minimize ongoing costs and operational complexity over the product lifecycle.
What should companies look for in a partner to develop an auto-injector?
When selecting a partner to develop an auto-injector, companies should look for an organization with deep, end-to-end experience across drug delivery device design, development and manufacturing.
A strong partner not only brings these end-to-end capabilities but also complements your existing team and helps fill critical gaps needed to reach your goals. This may include a head start through proven platform technologies like the A.i.r. Platform™ and the Intellectual Property (IP) behind it, as well as the expertise required to develop and scale a final device.
A partner like MGS is able to support the full lifecycle of the device from early design optimization and incorporation of usability insights, through tooling development, automation integration and scalable manufacturing. This continuity helps ensure that design decisions made early in development translate reliably into production.
Importantly, that support can extend beyond device development into the next phase, where the device is combined with the drug. Experience in this area helps ensure a more seamless transition and reduces risk as programs move toward commercialization.
Working with a single, experienced manufacturing partner across these stages can help reduce handoffs, minimize development risk and support a more efficient path to commercialization.
This type of integrated platform-based development approach is reflected in offerings such as the A.i.r. Platform™ from MGS.

Want to learn more about the A.i.r. Platform™?
From Design & Development to Tooling, Automation and Manufacturing, our experts have the insights you need to accelerate your auto-injector innovations. Send us a message today to start your drug delivery device development!
